Introduction
Hyperuricemia has emerged as a common biochemical abnormality in parallel with the global rise in obesity, type 2 diabetes, and metabolic syndrome. Increasing epidemiological evidence indicates that both symptomatic hyperuricemia, manifesting as gout, and asymptomatic elevations in serum uric acid are highly prevalent in industrialized and rapidly urbanizing societies. In many cohorts, a substantial proportion of individuals with metabolic syndrome exhibit uric acid concentrations above traditionally accepted “normal” ranges, even in the absence of overt gout. This clustering suggests that dysregulated uric acid metabolism is closely intertwined with the broader milieu of insulin resistance, visceral adiposity, and low‑grade inflammation that characterizes modern metabolic disease.
Beyond its classical role in gout, uric acid is now recognized as more than a passive biomarker of disease burden. Experimental and clinical data increasingly support a contributory role of elevated uric acid in the pathogenesis of hypertension, chronic kidney disease (CKD), non‑alcoholic fatty liver disease (NAFLD), and cardiovascular disease. Uric acid can promote oxidative stress, endothelial dysfunction, and activation of the renin–angiotensin–aldosterone system, thereby facilitating the development of elevated blood pressure and vascular injury. In the kidney, sustained hyperuricemia has been linked to glomerular hypertension, tubulointerstitial damage, and progressive decline in renal function, while in the liver, it is associated with hepatocellular fat accumulation and progression of NAFLD. Collectively, these observations position uric acid as both a clinically accessible biomarker and a potential causal mediator across multiple cardio‑renal‑metabolic domains.
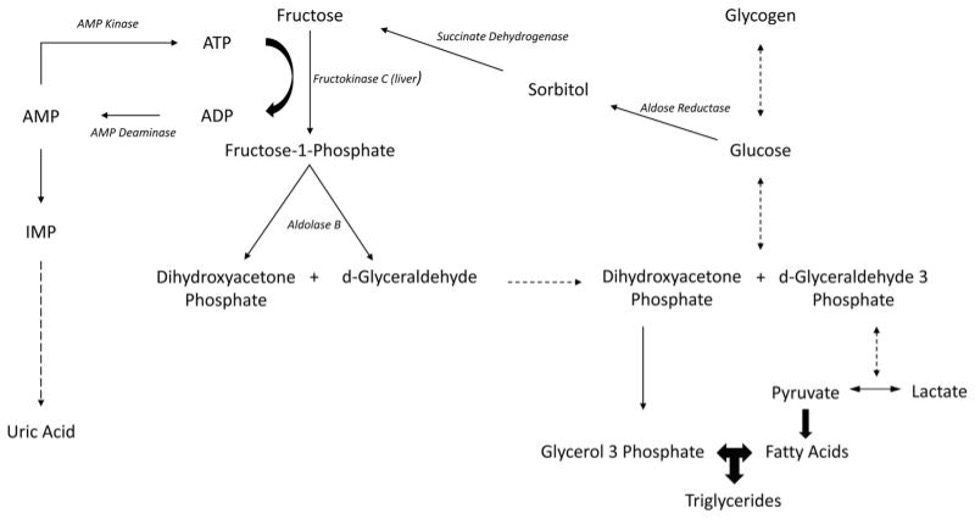
A central dietary driver of hyperuricemia is excessive fructose intake, particularly in the form of added sugars. Fructose undergoes rapid hepatic phosphorylation, leading to accelerated ATP degradation, accumulation of AMP, and subsequent conversion of AMP to uric acid. This biochemical pathway can acutely raise serum uric acid levels and, when chronically engaged, contributes to sustained hyperuricemia and downstream metabolic derangements. In parallel, fructose metabolism promotes de novo lipogenesis, hepatic fat accumulation, and insulin resistance, reinforcing the tight pathophysiological coupling between uric acid excess and metabolic dysfunction.
Modern dietary patterns, characterized by frequent consumption of sugar‑sweetened beverages and highly processed foods, provide the ideal substrate for activating this fructose–uric acid axis. Soft drinks, fruit juices, sweetened teas and coffees, as well as packaged snacks and desserts, are major contributors of added fructose in many populations. Conventional lifestyle advice for individuals with hyperuricemia or metabolic syndrome typically emphasizes reducing intake of sugar‑sweetened beverages and refined carbohydrates. While this strategy is mechanistically sound, long‑term adherence can be challenging in real‑world settings where sweet taste is deeply embedded in cultural and social practices. This adherence gap has driven growing interest in alternative sweeteners that can reduce the glycemic and fructose load without exacerbating uric acid metabolism, creating a rationale to explore rare sugars such as allulose within uric‑acid‑conscious, preventive nutrition frameworks.
Pathophysiology of Uric Acid in Metabolic Health
Uric acid is the final oxidation product of purine catabolism in humans and higher primates, reflecting both endogenous nucleotide turnover and exogenous purine intake. Purine nucleotides are degraded to hypoxanthine and xanthine, which are sequentially oxidized by xanthine oxidoreductase, present as xanthine dehydrogenase and xanthine oxidase to generate uric acid. Xanthine oxidase is highly expressed in the liver and intestinal mucosa, and its activity not only determines uric acid production but also generates reactive oxygen species that can contribute to oxidative stress. In humans, the lack of uricase, which further degrades uric acid to allantoin in most other mammals, results in higher baseline serum uric acid concentrations compared with species that retain this enzyme. Once produced, approximately 70% of urate is excreted by the kidneys and the remaining 30% via the intestine, with serum levels determined by the balance between production and excretion. Hyperuricemia therefore arises from either increased generation of uric acid (for example, high purine or fructose load, high xanthine oxidase activity) or reduced renal and intestinal clearance, with defective elimination accounting for the majority of clinical cases [1,2,3,4,5,6,7,8].

Fructose metabolism provides a key biochemical link between diet, ATP depletion, and uric acid generation. After ingestion, fructose is rapidly phosphorylated by fructokinase in the liver, consuming ATP and inorganic phosphate in a reaction that lacks effective negative feedback, which leads to transient intracellular phosphate depletion and ATP fall. This energetic stress activates AMP deaminase, promoting the degradation of AMP to inosine monophosphate and downstream metabolites that are ultimately oxidized to uric acid, causing an acute rise in intracellular and circulating uric acid levels. Repeated high fructose loads chronically engage this pathway, sustaining hyperuricemia and simultaneously stimulating de novo lipogenesis, hepatic triglyceride accumulation, and impairment of fatty acid oxidation. Uric acid itself can further exacerbate mitochondrial oxidative stress and impair hepatic energy status, reinforcing a vicious cycle that promotes non‑alcoholic fatty liver disease (NAFLD) and systemic insulin resistance. In parallel, fructose‑induced increases in uric acid contribute to endothelial dysfunction and inflammation, recognized early steps in vascular injury and the broader cardiorenal metabolic syndrome [9,10,11,12].
A growing body of epidemiological and experimental evidence links elevated uric acid with a spectrum of metabolic and cardiorenal disorders, including insulin resistance, hypertension, NAFLD, chronic kidney disease (CKD), and cardiovascular disease (CVD). Observational studies consistently report higher prevalence of hyperuricemia among individuals with metabolic syndrome, type 2 diabetes, and combined CKD–NAFLD, and identify serum uric acid as an independent predictor of renal function decline and adverse cardiovascular outcomes. Mechanistically, uric acid can induce endothelial dysfunction, activate the renin–angiotensin–aldosterone system, and promote vascular smooth muscle proliferation, thereby contributing to hypertension and atherosclerotic risk, while in the kidney it is associated with glomerular hypertension, tubulointerstitial injury, and accelerated CKD progression. Interventional data suggest that pharmacologic urate lowering may improve insulin resistance and systemic inflammation in hyperuricemic individuals, supporting a potential causal role; however, reverse causality and shared risk factors such as obesity and dyslipidaemia also contribute, making the relationship bidirectional and complex. Taken together, these findings position uric acid not only as a marker of gout risk but as a modifiable node within interconnected metabolic, vascular, and renal networks, and therefore a relevant therapeutic target in the broader context of cardio‑renal‑metabolic disease [6,13,14,15].
Allulose: Chemistry, Metabolism and Safety
Allulose, also known as D‑allulose or D‑psicose, is a C‑3 epimer of D‑fructose and is classified as a rare monosaccharide that occurs in small amounts in nature. It provides approximately 70% of the sweetness of sucrose yet contributes less than 0.4 kcal/g, largely because it is poorly metabolized after absorption. Human toxicokinetic studies indicate that roughly 70% of ingested allulose is absorbed in the small intestine via fructose transporters, appears in the circulation largely unchanged, and is rapidly excreted in the urine, while the remaining fraction passes into the colon and is excreted in feces with only limited fermentation by gut microbiota. This combination of poor utilization as an energy substrate and rapid renal elimination underpins the minimal caloric contribution and low glycemic impact of allulose, as it enters glycolytic and lipogenic pathways to only a very limited extent compared with glucose or fructose [16,17,18,19,20,21].
The low glycemic profile of allulose is supported by controlled human trials and quantitative syntheses. A systematic review and meta‑analysis of intervention studies in healthy adults demonstrated that co‑ingestion of 5–10 g of allulose with a carbohydrate‑containing meal significantly reduced the incremental area under the curve for postprandial blood glucose compared with the same meal without allulose, and also attenuated postprandial insulin excursions. In individuals with type 2 diabetes, a more recent meta‑analysis similarly reported that allulose intake reduced postprandial glucose levels and time above target range, supporting its role as a dietary adjunct for glycemic management. In preclinical models of diet‑induced obesity and type 2 diabetes, 8–12 weeks of dietary allulose supplementation consistently improved fasting glucose, insulin resistance indices (including HOMA‑IR), glucose tolerance, and body weight or adiposity when compared with high‑fat or Western‑style diets alone. Mechanistic work in rodents suggests that these effects are mediated by enhanced hepatic insulin sensitivity with down‑regulation of gluconeogenic enzymes, modulation of intestinal sugar absorption, activation of AMPK pathways, improved hepatic and skeletal muscle lipid handling, increased postprandial fat oxidation, and anti‑inflammatory effects in liver and adipose tissue [17,19,21,22,23,24,25,26].
Human safety data to date indicate that allulose is generally well tolerated and metabolically neutral or beneficial when consumed within studied doses. Acute crossover studies comparing intragastric administration of allulose with tap water or other sweeteners have not shown adverse effects on plasma lipids, inflammatory markers, or serum uric acid. Longer‑term trials, including a 12‑week study in which participants consumed 5 g of D‑allulose with each meal three times daily, reported no clinically significant changes in standard hematology, liver and renal biochemistry, or uric acid concentrations, suggesting at least uric‑acid neutrality over the duration studied. Additionally, a 48‑week trial in individuals with elevated LDL cholesterol found that daily allulose intake did not worsen lipid profiles or high‑sensitivity C‑reactive protein, further supporting its cardiovascular and inflammatory safety. Gastrointestinal symptoms such as bloating, diarrhea, or abdominal discomfort can occur at higher single doses or with rapid dose escalation, reflecting osmotic and fermentative effects, but these events are generally mild, dose‑dependent, and mitigated by distributing intake across meals. Overall, the available evidence supports allulose as a low‑calorie, low‑glycemic rare sugar with favourable effects on glycemic control and weight regulation, and a reassuring short‑ to medium‑term safety profile that appears neutral with respect to serum uric acid [17,18,19,27].
Evidence for Effects of Allulose on Uric Acid
Controlled human data on allulose and uric acid remain limited but are consistent in showing a neutral effect on serum uric acid, while mechanistic and preclinical work suggests plausible urate‑modulating pathways that still require confirmation.
Human Experimental Data
In a randomized, double‑blind, crossover study of 18 healthy adults, acute intragastric administration of 25 g D‑allulose was compared with 50 g erythritol and 300 mL tap water, with serial measurements of glucose, lipids, inflammatory markers, and uric acid. Across the observation period, D‑allulose did not produce any significant change in serum uric acid concentrations relative to tap water, indicating an acutely neutral effect on urate despite a substantially higher osmotic load than in usual dietary use. Importantly, the study was explicitly designed to assess safety parameters, and the absence of uric‑acid elevation was interpreted as supportive of allulose’s metabolic safety profile [27].
Longer‑term data are available from a 12‑week intervention in which adults consumed 5 g D‑allulose in tea three times daily with meals, compared with a sucrose control beverage of similar sweetness. In this trial, daily allulose intake did not significantly alter serum uric acid concentrations over 12 weeks, and urate values remained comparable to those in the sucrose group, suggesting that replacing sucrose with allulose neither raises nor lowers uric acid over the short‑ to medium‑term in otherwise healthy individuals. A separate 48‑week study in people with elevated LDL cholesterol similarly reported no adverse changes in uric acid alongside neutral effects on lipids and high‑sensitivity C‑reactive protein, reinforcing a picture of urate neutrality rather than urate lowering in current human datasets. Collectively, these controlled trials do not demonstrate a clear uric‑acid lowering effect of allulose; instead, they support its safety and the absence of uric acid elevation when used acutely or chronically at typical supplemental doses [18,27].
Preclinical and Mechanistic Hypotheses
Despite the neutral uric acid trajectories observed in short‑term human trials, several mechanistic pathways suggest how chronic allulose use could favour urate handling indirectly. In rodent models of diet‑induced obesity and type 2 diabetes, sustained allulose supplementation improves insulin sensitivity, reduces hepatic steatosis, and attenuates visceral adiposity, in part via activation of AMPK, modulation of adipogenic transcription factors, and improved hepatic and muscle lipid metabolism. Because insulin resistance is tightly linked to renal urate retention through effects on renal hemodynamics and urate transporters, amelioration of insulin resistance and NAFLD by allulose could secondarily enhance uric acid excretion over time, even if allulose does not directly inhibit uric acid production [13,15,19,26,28,29].
Expert commentary and translational discussions have proposed more direct effects of allulose on renal urate handling, particularly via modulation of urate transporters in the proximal tubule. For example, clinicians such as Bikman and colleagues have hypothesized that allulose may up‑regulate efflux transporters (e.g., ABCG2, NPT4) and/or down‑regulate reabsorptive transporters (e.g., URAT1, GLUT9), thereby enhancing renal uric acid excretion and lowering serum levels. Other speculative mechanisms include competition with fructose for intestinal and renal transport, partial blockade of fructose‑induced ATP depletion and uric acid generation in the liver, and an AMPK‑driven shift toward greater fat and glucose oxidation that could reduce the purine‑catabolic pressure leading to urate formation. However, these mechanisms remain largely inferential: they are extrapolated from known transporter biology, AMPK signalling data, and improvements in metabolic phenotype, rather than from direct human studies that quantify urate transport, fractional excretion of urate, or transporter expression in response to allulose. Thus, while biologically plausible and internally consistent with allulose’s broader metabolic effects, these hypotheses have not yet been rigorously tested in mechanistic human trials with uric acid as a primary endpoint [14,19,26,27,28,29,30].
Clinical Commentary and Real-World Observations
In parallel with formal research, metabolic clinicians and educators have begun to feature allulose as a preferred sweetener for hyperuricemic or metabolically compromised patients, emphasizing its ability to provide sweetness without fructose‑driven uric acid production and with added benefits for glycemic control and body composition. Podcast discussions and professional talk, such as those by Bikman and by clinicians focused on low‑carbohydrate and keto interventions often describe reductions in serum uric acid observed in individual patients after substituting allulose for conventional sugars, alongside improvements in insulin resistance and weight. These accounts frequently reference animal data, transporter biology, and early human safety studies to argue that allulose is unlikely to exacerbate hyperuricemia and may, in some individuals, contribute to uric acid reduction [19,26,27].
However, such clinical observations and expert opinions remain hypothesis‑generating rather than definitive evidence. They are typically derived from uncontrolled, multifaceted lifestyle interventions where weight loss, reduced fructose intake, and improved diet quality all co‑occur with allulose use, making it impossible to attribute uric acid changes specifically to allulose. At present, no large randomized controlled trials have been published with serum uric acid, urinary uric acid excretion, or gout flares as primary outcomes comparing allulose to fructose, sucrose, or non‑nutritive sweeteners. As such, clinical enthusiasm for allulose as a uric‑acid‑lowering agent should be tempered by the recognition that robust interventional evidence is lacking; current data justify positioning allulose as a metabolically favourable, uric‑acid‑neutral sugar substitute, while its potential as a targeted urate‑modulating therapy remains an important but unproven research question [13,14,15,19,27].
Integrating Allulose into a Uric Acid-Conscious Lifestyle Framework
Integrating allulose into a uric-acid-conscious lifestyle framework is best done as part of a comprehensive metabolic protocol, rather than as a stand-alone urate-lowering therapy.
Dietary strategies beyond Pharmacology
Lifestyle guidance for hyperuricemia and gout now emphasizes reducing fructose‑rich sugars, moderating purine intake, achieving weight loss, and adopting plant‑forward patterns such as Mediterranean or DASH‑style diets. Excess fructose from sugar‑sweetened beverages and processed foods is a major driver of hyperuricemia, so recommendations increasingly focus on replacing these with water or unsweetened beverages, choosing whole fruit instead of fruit juice, and limiting added sugars. Allulose can be positioned as one tool within this broader protocol: substituting it for sucrose or high‑fructose corn syrup reduces both glycemic load and fructose exposure while maintaining palatability, and current data indicate that such substitution does not increase serum uric acid. In parallel, core elements such as weight reduction, increased physical activity, higher fiber intake, and more vegetables, legumes, and whole grains remain central for improving insulin sensitivity, NAFLD, and urate handling [18,19,27,31,32,33,34,35,36].
From a preventive and biohacking perspective, allulose fits as an adjunct in a “low‑fructose, low‑purine, high‑fiber” pattern that also prioritizes hydration, moderation of alcohol (especially beer and spirits), and individualized energy restriction to support fat loss in insulin‑resistant, hyperuricemic phenotypes. Within this context, the key message is that swapping conventional sugar for allulose can reduce caloric and glycemic burden and avoid fructose‑driven uric acid production, with the added advantage of short‑term uric‑acid neutrality demonstrated in human trials [18,19,27,31,34,35,36].
Practical Applications in Metabolic Clinics and Digital Health
In clinical practice, allulose can be incorporated into beverages and functional foods targeted to patients with metabolic syndrome, NAFLD, and prediabetes who require stringent glycemic and uric‑acid management but struggle with complete sugar avoidance. Examples include using allulose‑sweetened teas or coffees instead of sugar‑sweetened versions, reformulating yogurts or high‑protein snacks with allulose, or recommending commercially available allulose‑containing products as upgrades from sucrose‑rich options. For individuals with NAFLD, where lifestyle modification is the primary therapy, replacing fructose‑containing sweeteners with allulose can complement caloric restriction and increased physical activity to improve hepatic steatosis while minimizing uric‑acid stress [18,19,23,31,33,35].
Digital health programs that combine CGM, connected scales, and laboratory tracking provide a natural environment to operationalize this strategy. AI‑supported platforms have shown that integrating CGM with behavioral data can improve glycemic control and weight through personalized feedback and food‑specific guidance. Within such systems, algorithmic nudges can recommend allulose‑containing recipes or products when a user’s data show high postprandial glucose excursions to sugary foods, elevated BMI or waist circumference, and documented hyperuricemia. Over time, the platform can correlate shifts toward allulose and reduced fructose intake with trends in CGM metrics, body composition, and serum uric acid, enabling iterative personalization and hypothesis‑generating real‑world data on allulose’s urate impact [19,27,37,38].
Considerations and Cautions
Several practical cautions are important when integrating allulose into a uric‑acid‑conscious protocol. First, total sweetener exposure should be monitored: while allulose appears safe and non‑glycemic, excessive reliance on intense sweetness may perpetuate hedonic sugar seeking and displace whole foods; guidelines for hyperuricemia still emphasize overall dietary quality, weight loss, and moderation rather than simple sweetener substitution. Second, gastrointestinal tolerance varies; higher single doses of allulose can cause bloating, loose stools, or discomfort in some individuals, so clinicians should titrate doses, encourage divided intake with meals, and personalize recommendations based on symptoms. Third, while mechanistic and preclinical data suggest that improving insulin resistance and hepatic steatosis with allulose may secondarily enhance urate handling, and expert commentary has proposed more direct effects on renal urate transport, current human trials have not demonstrated a robust uric‑acid‑lowering effect [18,26,27,28,30,31,34,35,36].
Consequently, until randomized controlled trials are conducted with uric acid endpoints as primary outcomes, allulose should be framed as a metabolically favourable, uric‑acid‑neutral sugar substitute within comprehensive lifestyle therapy, rather than as a stand‑alone uric‑acid‑lowering agent. Pharmacologic urate‑lowering therapy, structured dietary counselling, weight management, and physical activity remain the evidence‑based cornerstones of hyperuricemia and gout management, with allulose functioning as a supportive nutritional tool that aligns well with modern preventive and biohacking strategies [19,27,31,34,35,36].
Research Gaps and Future Directions
Despite promising metabolic data, the evidence base for allulose as a uric‑acid‑modulating intervention remains preliminary, and several key research gaps must be addressed before it can be positioned beyond its current role as a metabolically favourable sugar substitute.
Primary Endpoint Trials
Most human allulose trials have focused on postprandial glycemia, body weight, and short‑term safety, with uric acid measured only as a secondary or exploratory outcome. Future randomized controlled trials are needed in which serum uric acid, urinary uric acid excretion, and clinically relevant endpoints such as gout flares are designated as primary outcomes when comparing allulose with sucrose, fructose, and non‑caloric sweeteners. Such studies should be adequately powered, of at least 6–12 months’ duration, and incorporate standardized dietary backgrounds to isolate the specific contribution of allulose, ideally including dose–response arms to determine whether higher habitual intakes exert differential effects on urate homeostasis [17,19,27,39,40].
Mechanistic Work
At the mechanistic level, available data suggest that allulose modulates hepatic and renal energy metabolism, activates AMPK, and influences purine‑related metabolites in animal models, but detailed human mechanistic work is lacking. Future studies should examine the impact of allulose on renal urate transporters (e.g., URAT1, GLUT9, ABCG2, OAT family) using a combination of fractional excretion measurements, transporter expression or activity assays, and, where feasible, renal imaging or biopsy‑derived biomarkers. Parallel investigations should evaluate whether allulose alters xanthine oxidase activity or purine turnover in vivo, and how its known AMPK‑activating and lipid‑modulating effects translate into changes in hepatic and renal purine metabolism. Carefully designed human mechanistic trials, using stable isotope tracers, metabolomics, and tissue‑specific imaging are needed to move beyond indirect inference from rodent models and establish causal pathways connecting allulose intake to urate production and excretion [3,14,22,24,26,28,30,41].
Phenotype-Specific Responses
Hyperuricemia occurs across heterogeneous clinical phenotypes, including gout, chronic kidney disease, metabolic syndrome, and so‑called “lean hyperuricemia,” each with distinct pathophysiology and therapeutic needs. Future allulose research should therefore employ stratified designs that enroll and analyze these groups separately, rather than pooling them into broad “at‑risk” populations. In patients with gout, trials could assess whether replacing fructose‑containing sugars with allulose reduces flare frequency or urate burden when added to standard urate‑lowering therapy; in CKD, studies must consider altered urate and allulose handling and carefully monitor renal function and safety. In metabolic syndrome and NAFLD, where insulin resistance and hepatic steatosis are central, it will be important to determine whether improvements in these parameters with allulose use translate into disproportionate benefits for uric acid compared with other sweeteners. Finally, targeted studies in lean individuals with hyperuricemia could help clarify whether allulose’s potential urate effects are contingent on insulin resistance and adiposity, or extend to phenotypes where purine overproduction and genetic variants in urate transporters predominate [13,14,15,23,30,33,35].
Long-term Safety
Current human safety data for allulose include acute and short‑ to medium‑term interventions (up to 48 weeks), which support a favourable profile and uric‑acid neutrality but do not fully address lifetime exposure or high‑frequency consumption patterns. Regulatory reviews in Europe and national risk assessments have also highlighted outstanding uncertainties, including gaps related to proposed use levels, genotoxicity, and potential effects on the gut microbiome and pathogenic bacteria, and have called for additional human data before broad approval. Future work should therefore include multi‑year surveillance studies or pragmatic trials that track individuals with high habitual allulose intake, often in combination with other rare sugars and sugar alcohols, assessing not only metabolic endpoints and serum uric acid but also renal function, microbiome composition, and potential off‑target effects. These efforts will be particularly important as allulose‑containing products proliferate and cumulative exposure increases at a population level [18,27,42].
For the purposes of a conceptual figure, this section can be visually summarized by contrasting two pathways: (1) fructose intake leading to ATP depletion, increased uric acid production, oxidative stress, NAFLD, insulin resistance, and heightened cardio‑renal risk; versus (2) allulose intake, which based on current evidence is hypothesized to maintain neutral or reduced uric acid exposure while improving glycemic control, adiposity, and broader metabolic health, pending confirmation from the targeted trials and mechanistic studies outlined above [9,19,26,27].
Conclusion
Allulose has emerged as a rare sugar with robust evidence for attenuating postprandial glycemic excursions and supporting weight management, while demonstrating a favourable short‑ to medium‑term safety profile that includes neutral effects on serum uric acid in the available human trials. Within the broader context of cardio‑renal‑metabolic disease, this combination of low caloric value, minimal glycemic impact, and uric‑acid neutrality positions allulose as an attractive alternative to conventional caloric sweeteners.
Given the strong pathophysiological links between excess fructose intake, ATP depletion, uric acid overproduction, and downstream metabolic dysfunction, preferential use of allulose instead of sucrose or high‑fructose corn syrup represents a rational, low‑risk nutritional intervention for individuals with metabolic syndrome, NAFLD, or elevated uric acid. By reducing both glycemic and fructose loads without sacrificing palatability, allulose can help close the adherence gap often observed with strict sugar‑restriction strategies, while avoiding the uric‑acid‑raising potential inherent to fructose‑containing sweeteners.
At the same time, early mechanistic work and expert commentary suggesting that allulose may enhance renal uric acid excretion and function as a urate‑modulating sweetener must be interpreted with appropriate caution. These hypotheses, grounded in improvements in insulin sensitivity, hepatic steatosis, and energy metabolism, remain insufficiently tested in human studies specifically designed with uric acid–related endpoints. Rigorous randomized trials, incorporating serum and urinary uric acid, gout flares, and transporter‑level mechanistic readouts are needed before allulose can be credibly positioned as a dedicated uric-acid-lowering therapy rather than as a uric‑acid‑neutral sugar substitute with broader metabolic benefits.
In the era of AI‑enabled, personalized preventive medicine, allulose can be integrated into multi‑modal care programs that combine real‑time monitoring of glycemia, body composition, and uric acid with adaptive digital coaching. Within such frameworks, algorithm‑driven recommendations can support targeted replacement of conventional sugars with allulose in individuals displaying concurrent hyperuricemia and metabolic risk, while longitudinal data streams help refine individual response profiles. This systems‑level approach offers a promising pathway by which allulose can contribute to sustainable lifestyle change, attenuate metabolic and urate‑related risk, and help bridge the gap between mechanistic insight and real‑world prevention
Reference
- Maiuolo J, Oppedisano F, Gratteri S, Muscoli C, Mollace V. Regulation of uric acid metabolism and excretion. International Journal of Cardiology [Internet]. 2016 Jun;213:8–14. Available from: https://www.sciencedirect.com/science/article/pii/S0167527315303429
- Barr WG. Uric Acid [Internet]. Nih.gov. Butterworths; 2014. Available from: https://www.ncbi.nlm.nih.gov/books/NBK273/
- Hyndman D, Liu S, Miner JN. Urate Handling in the Human Body. Current Rheumatology Reports. 2016 Apr 22;18(6).
- Furuhashi M. New insights into purine metabolism in metabolic diseases: role of xanthine oxidoreductase activity. American Journal of Physiology-Endocrinology and Metabolism. 2020 Nov 1;319(5):E827–34.
- Uric Acid – an overview | ScienceDirect Topics [Internet]. www.sciencedirect.com. Available from: https://www.sciencedirect.com/topics/pharmacology-toxicology-and-pharmaceutical-science/uric-acid
- Park JH, Jo YI, Lee JH. Renal effects of uric acid: hyperuricemia and hypouricemia. The Korean Journal of Internal Medicine. 2020 Nov 1;35(6):1291–304.
- Lohr J. Hyperuricemia: Practice Essentials, Pathophysiology, Etiology. eMedicine [Internet]. 2021 Apr 3; Available from: https://emedicine.medscape.com/article/241767-overview
- Uric Acid Metabolism and Associated Health Impacts | Nature Research Intelligence [Internet]. Nature.com. 2023. Available from: https://www.nature.com/research-intelligence/nri-topic-summaries/uric-acid-metabolism-and-associated-health-impacts-micro-1117
- Jensen T, Abdelmalek MF, Sullivan S, Nadeau KJ, Green M, Roncal C, et al. Fructose and sugar: A major mediator of non-alcoholic fatty liver disease. Journal of Hepatology [Internet]. 2018 May 1;68(5):1063–75. Available from: https://pubmed.ncbi.nlm.nih.gov/29408694/
- Greene HL, Wilson FA, Hefferan P, Terry AB, Moran JR, Slonim AE, et al. ATP depletion, a possible role in the pathogenesis of hyperuricemia in glycogen storage disease type I. The Journal of clinical investigation [Internet]. 1978 Aug;62(2):321–8. Available from: https://pubmed.ncbi.nlm.nih.gov/276529/
- Jia G, Aroor AR, Whaley-Connell AT, Sowers JR. Fructose and Uric Acid: Is There a Role in Endothelial Function? Current Hypertension Reports. 2014 Apr 24;16(6).
- Russo E, Leoncini G, Esposito P, Garibotto G, Pontremoli R, Viazzi F. Fructose and Uric Acid: Major Mediators of Cardiovascular Disease Risk Starting at Pediatric Age. International Journal of Molecular Sciences. 2020 Jun 24;21(12):4479.
- Golmohammadi S, Tavasoli M, Asadi N. Prevalence and Risk Factors of Hyperuricemia in Patients with Chronic Kidney Disease and Non-Alcoholic Fatty Liver. Clinical and Experimental Gastroenterology. 2020 Aug;Volume 13:299–304.
- Du L, Zong Y, Li H, Wang Q, Xie L, Yang B, et al. Hyperuricemia and its related diseases: mechanisms and advances in therapy. Signal Transduction and Targeted Therapy [Internet]. 2024 Aug 28;9(1). Available from: https://www.nature.com/articles/s41392-024-01916-y
- Johnson RJ, Bakris GL, Borghi C, Chonchol MB, Feldman D, Lanaspa MA, et al. Hyperuricemia, Acute and Chronic Kidney Disease, Hypertension, and Cardiovascular Disease: Report of a Scientific Workshop Organized by the National Kidney Foundation. American Journal of Kidney Diseases [Internet]. 2018 Jun;71(6):851–65. Available from: https://www.ajkd.org/article/S0272-6386(18)30087-8/pdf
- The Declaration of Allulose and Calories from Allulose on Nutrition and Supplement Facts Labels: Guidance for Industry [Internet]. Available from: https://www.fda.gov/media/123342/download
- Tani Yuma, Tokuda M, Nishimoto N, Yokoi H, Izumori K. Allulose for the attenuation of postprandial blood glucose levels in healthy humans: A systematic review and meta-analysis. PLOS ONE. 2023 Apr 6;18(4):e0281150–0.
- Supporting document 1 (At Approval) [Internet]. 2024 [cited 2026 Feb 17]. Available from: https://www.foodstandards.gov.au/sites/default/files/2024-08/A1247_SD1_at_Approval.pdf
- Xia Y, Cheng Q, Mu W, Hu X, Sun Z, Qiu Y, et al. Research Advances of d-allulose: An Overview of Physiological Functions, Enzymatic Biotransformation Technologies, and Production Processes. Foods. 2021 Sep 15;10(9):2186.
- Hossain A, Yamaguchi F, Matsuo T, Tsukamoto I, Toyoda Y, Ogawa M, et al. Rare sugar d-allulose: Potential role and therapeutic monitoring in maintaining obesity and type 2 diabetes mellitus. Pharmacology & Therapeutics. 2015 Nov;155:49–59.
- Kimura T, Kanasaki A, Hayashi N, Yamada T, Iida T, Nagata Y, et al. d-Allulose enhances postprandial fat oxidation in healthy humans. Nutrition. 2017 Nov;43-44:16–20.
- Cayabyab KB, Shin MJ, Heimuli MS, Kim IJ, D’Agostino DP, Johnson RJ, et al. The Metabolic and Endocrine Effects of a 12-Week Allulose-Rich Diet. Nutrients [Internet]. 2024 Jan 1;16(12):1821. Available from: https://www.mdpi.com/2072-6643/16/12/1821
- Kamal S, Gou Y, Tsuzuki T, Fu L, Yamada T, Banno R, et al. d-Allulose Ameliorates Fructose-Induced Skeletal Muscle Insulin Resistance via Regulation of Ectopic Lipid Accumulation Independent of Body Weight Changes. Nutrients [Internet]. 2025;17(12):2050. Available from: https://pubmed.ncbi.nlm.nih.gov/40573161/
- Akane Kanasaki, Misato Niibo, Iida T. Metabolic Profiling of Rat Kidney Tissue Following Administration of D-Allulose. Journal of Applied Glycoscience. 2024 Jun 21;71(3):73–80.
- Ayesh H, Suhail S, Ayesh S. Impact of allulose on blood glucose in type 2 diabetes: A meta-analysis of clinical trials. Metabolism Open [Internet]. 2024 Dec;24:100329. Available from: https://meddocsonline.org/journal-of-psychiatry-and-behavioral-sciences/perinatal-asphyxia-attention-deficit-hyperactivity-disorder-a-retrospective.pdf
- Han Y, Kwon EY, Choi MS. Anti-Diabetic Effects of Allulose in Diet-Induced Obese Mice via Regulation of mRNA Expression and Alteration of the Microbiome Composition. Nutrients. 2020 Jul 16;12(7):2113.
- Teysseire F, Bordier V, Budzinska A, Van Oudenhove L, Weltens N, Beglinger C, et al. Metabolic Effects and Safety Aspects of Acute D-allulose and Erythritol Administration in Healthy Subjects. Nutrients. 2023 Jan 15;15(2):458.
- Lee GH, Peng C, Lee HY, Park SA, Hoang TH, Kim JH, et al. D-allulose ameliorates adiposity through the AMPK-SIRT1-PGC-1α pathway in HFD-induced SD rats. Food & Nutrition Research. 2021 Dec 21;65.
- Tsuzuki T, Suzuki R, Kajun R, Yamada T, Iida T, Liu B, et al. Combined effects of exercise training and D‐allulose intake on endurance capacity in mice. Physiological Reports [Internet]. 2022 May 1 [cited 2025 Apr 10];10(9). Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC9095992/
- Yang B, Xin M, Liang S, Xu X, Cai T, Dong L, et al. New insight into the management of renal excretion and hyperuricemia: Potential therapeutic strategies with natural bioactive compounds. Frontiers in Pharmacology. 2022 Nov 22;13.
- Lubawy M, Formanowicz D. High-Fructose Diet–Induced Hyperuricemia Accompanying Metabolic Syndrome–Mechanisms and Dietary Therapy Proposals. International Journal of Environmental Research and Public Health. 2023 Feb 17;20(4):3596.
- Checking your browser – reCAPTCHA [Internet]. Nih.gov. 2024 [cited 2026 Feb 17]. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC8996397/
- Montemayor S, Bouzas C, Mascaró CM, Casares M, Llompart I, Abete I, et al. Effect of Dietary and Lifestyle Interventions on the Amelioration of NAFLD in Patients with Metabolic Syndrome: The FLIPAN Study. Nutrients. 2022 May 26;14(11):2223.
- Dietary Guide for Hyperuricemia and Gout Patients (WS/T 560-2017). PubMed. 2023 Sep 20;36(9):897–8.
- Ahmed IA, Mikail MA, Mustafa MR, Ibrahim M, Othman R. Lifestyle interventions for non-alcoholic fatty liver disease. Saudi Journal of Biological Sciences. 2019 Jan;
- Nielsen SM, Zobbe K, Kristensen LE, Christensen R. Nutritional recommendations for gout: An update from clinical epidemiology. Autoimmunity Reviews. 2018 Nov;17(11):1090–6.
- Veluvali A, Dehghani Zahedani A, Hosseinian A, Aghaeepour N, McLaughlin T, Woodward M, et al. Impact of digital health interventions on glycemic control and weight management. npj Digital Medicine [Internet]. 2025 Jan 9;8(1). Available from: https://www.nature.com/articles/s41746-025-01430-7
- Ji C, Jiang T, Liu L, Zhang J, You L. Continuous glucose monitoring combined with artificial intelligence: redefining the pathway for prediabetes management. Frontiers in Endocrinology. 2025 May 26;16.
- Noronha JC, Braunstein CR, Glenn AJ, Khan TA, Viguiliouk E, Noseworthy R, et al. The effect of small doses of fructose and allulose on postprandial glucose metabolism in type 2 diabetes: A double‐blind, randomized, controlled, acute feeding, equivalence trial. Diabetes, Obesity and Metabolism. 2018 Jun 25;20(10):2361–70.
- gov. 2026 [cited 2026 Feb 17]. Available from: https://clinicaltrials.gov/study/NCT04027283
- Niibo M, Kanasaki A, Iida T, Ohnishi K, Ozaki T, Akimitsu K, et al. d-allulose protects against diabetic nephropathy progression in Otsuka Long-Evans Tokushima Fatty rats with type 2 diabetes. PloS one [Internet]. 2022;17(1):e0263300. Available from: https://pubmed.ncbi.nlm.nih.gov/35100325/
- Turck D, Montaña Cámara, Bohn T, Castenmiller J, Stefaan De Henauw, Ángeles Jos, et al. Safety of D‐allulose as a novel food pursuant to Regulation (EU) 2015/2283. EFSA Journal. 2025 Jun 1;23(6).