Introduction
Nicotinamide adenine dinucleotide (NAD+) is a ubiquitous pyridine nucleotide that functions both as an essential redox cofactor and as a substrate for multiple signaling enzymes that govern cellular stress responses, genomic maintenance, and metabolic adaptation. Traditionally viewed through the lens of energy metabolism as a key regulator of glycolysis, the tricarboxylic acid cycle, and oxidative phosphorylation, NAD+ is now recognized as a nodal integrator of pathways that connect nutrient availability, mitochondrial function, circadian biology, and inflammatory tone across organ systems. This expanded understanding has positioned NAD+ at the forefront of geroscience, with implications that extend from basic cellular bioenergetics to systems-level regulation of human health span.
A consistent observation across species is that NAD+ levels decline with aging, with further reductions observed in the context of cardiometabolic risk factors such as obesity, insulin resistance, and hypertension. Mechanistically, this decline reflects a complex interplay between reduced biosynthesis, particularly within the NAMPT-dependent salvage pathway and increased consumption by NAD+- dependent enzymes including poly (ADP-ribose) polymerases (PARPs), sirtuins, and the ectoenzyme CD38, which is upregulated with age and chronic inflammation. The resulting perturbation in NAD+ homeostasis has been linked to several hallmarks of aging, including mitochondrial dysfunction, impaired DNA repair, loss of proteostasis, altered nutrient sensing, and low-grade systemic inflammation.
Preclinical studies have shown that restoration of NAD+ levels through genetic and pharmacological means can ameliorate metabolic dysfunction, improve vascular and cardiac performance, enhance neuroprotective mechanisms, and delay the onset of multiple age-associated phenotypes. Interventions such as supplementation with NAD+ precursors (e.g., nicotinamide riboside and nicotinamide mononucleotide), activation of biosynthetic pathways, or inhibition of NAD+- consuming enzymes have been reported to improve insulin sensitivity, increase mitochondrial biogenesis, and extend health span in several animal models, although effects on maximal lifespan are more variable. These data have fueled interest in NAD+ as a therapeutic target in metabolic disease, cardiovascular aging, neurodegeneration, and regenerative medicine.
In parallel, early-phase human studies indicate that oral NAD+ precursors can safely increase circulating and tissue NAD+ levels, with emerging but still inconclusive evidence for benefits on cardiometabolic risk markers, vascular function, and self-reported fatigue or vitality. At the same time, the rapid commercialization of NAD+- boosting products and intravenous protocols has outpaced the strength of clinical evidence, raising important questions about long-term safety, appropriate dosing, and patient selection, particularly in population at risk for malignancy or with complex multimorbidity. These tensions underscore the need for a critical, evidence-based appraisal of NAD+ biology and its translational potential in. metabolic and aging wellness programs.
Thie review synthesizes current knowledge on NAD+ metabolism, its role in the biology of aging and metabolic regulation, and the existing clinical data on NAD+- boosting interventions, with a focus on application relevant to preventive, cardiometabolic, and regenerative health paradigms. By integrating mechanistic insights with human trial data and highlighting key uncertainties, the article aims to provide clinicians, researchers, and digital health practitioners with a balanced framework for incorporating NAD+- targeted strategies into health span-oriented care, while outlining for future research and responsible clinical translation.
NAD+ Biology and Metabolism
In mammalian cells, NAD+ homeostasis is maintained by three interconnected biosynthetic routes: the de novo pathway from tryptophan, the Preiss-Handler pathway from nicotinic acid, and, predominately, the nicotinamide (NAM) salvage pathway. The salvage pathway recycles NAM, generated by NAD+- consuming reactions, back to NAD+ via the rate-limiting enzyme nicotinamide phosphoribosyltransferase (NAMPT), which converts NAM to nicotinamide mononucleotide (NMN), followed by adenylation to NAD+ by NMN adenylyltransferases (NMNAT1-3). Additional precursors such as nicotinamide riboside (NR) and NMN enter this network by conversion to NMN through NR kinases (NRK1/2) or direct utilization, effectively bypassing the NAMPT step under some conditions and offering alternative routes to sustain NAD+ under stress, aging, or high-demand states. While de novo and preiss-Handler pathways contribute to NAD+ pools, especially in specific tissues such as liver and intestine, they appear to play a supporting role in most settings, with the salvage pathway supplying the majority of intracellular NAD+ required for continuous redox and signaling demands [1,2,3,4,5,6,7,8].
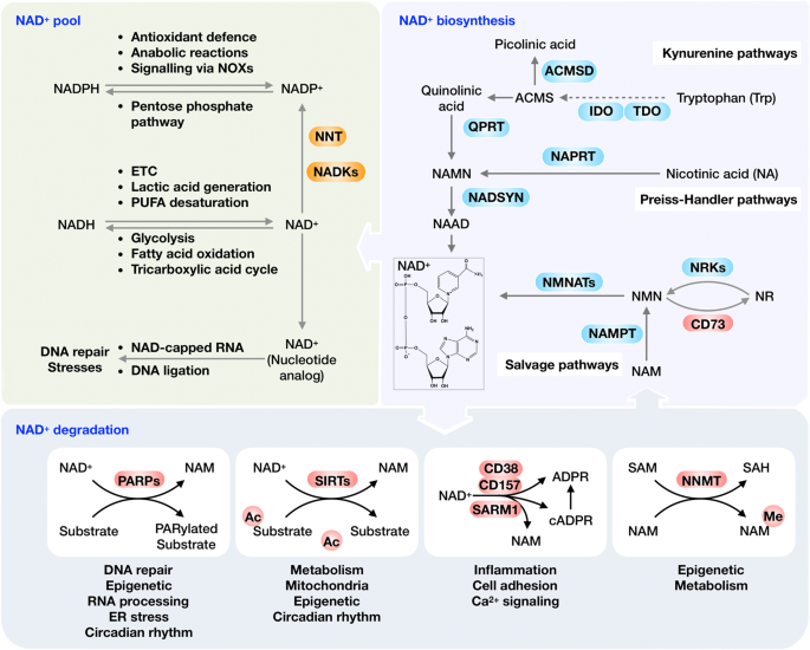
NAD+ is continually turned over by a set of NAD+- consuming enzymes that couple its cleavage to post-translational modification and second-messenger signaling, thereby linking metabolic state to genome maintenance, stress responses, and calcium signaling. Sirtuins (SIRT1-7) are NAD+- dependent deacylases and ADP-ribosyltransferases distributed across the nucleus, cytosol, and mitochondria, where they hydrolyze NAD+ to NAM and O-acetyl-ADP-ribose (or related ADP-ribose conjugates) to regulate chromatin structure, mitochondrial biogenesis, and metabolic adaptation to nutrient availability. Poly(ADP-ribose) polymerases (PARPs) use NAD+ to synthesize mono-or poly(ADP-ribose) chains on proteins and nucleic acids at sites of genotoxic stress, consuming large amounts of NAD+ during sustained DNA damage responses and thereby competing with sirtuins and other enzymes for the same intracellular NAD+ pool. CD38 and related ectoenzymes (CD157) act as NAD+ glycohydrolases and ADP-ribosyl cyclases, converting NAD+ to NAM, ADP-ribose, and cyclic ADP-ribose, potent calcium-mobilizing second messengers that modulate immune activation, survival, and metabolic signaling; with aging, CD38 expressing and activity increase and have been implicated as major drivers of NAD+ decline. Collectively, these enzymes form an integrated signaling network in which changes in NAD+ availability, as well as differential enzyme affinity for NAD+, dynamically prioritize DNA repair, stress resistance, and metabolic programs, and create therapeutic entry points at both the level of NAD+ production and targeted inhibition of high-flux NAD+ sinks such as PARP1 and CD38 [2,9,10,11,12].
NAD+ and the Biology of Aging
Links to the Hallmarks of Aging
The progressive decline in NAD+ levels during aging intersects with multiple established hallmarks of aging, positioning NAD+ metabolism as a central convergence point through which diverse cellular stressors influence biological aging trajectories. Reductions in NAD+ directly impair mitochondrial oxidative phosphorylation and compromise the capacity to maintain mitochondrial quality control via biogenesis, fusion-fission dynamics, and mitophagy, thereby accelerating mitochondrial dysfunction, one of the cardinal features of aging. NAD+ depletion also undermines proteostasis by limiting the activity of sirtuins and the mitochondrial unfolded protein response (UPR^mt^), leading to the accumulation of misfolded and aggregated proteins, defective organelles, and increased susceptibility to proteotoxic stress. Furthermore, because sirtuins function as NAD+- dependent sensors of metabolic state, low NAD+ availability blunts sirtuin-mediated regulation of AMPK, FOXO transcription factors, and other nutrient-sensing pathways, resulting in altered nutrient sensing and metabolic inflexibility that characterize aged cells and tissues [9,10,13,14,15,16,17,18].
By regulating sirtuin-mediated deacetylation and diacylation, NAD+ exerts pleiotropic effects on cellular resilience and adaptive capacity, most notably through the SIRT1-PGC-1a axis that governs mitochondrial biogenesis, fatty acid oxidation, gluconeogenesis, and antioxidant defenses. SIRT1 deacetylates and activates PGC-1a, which in turn induces transcription of nuclear-encoded mitochondrial genes and coordinates mitochondrial biogenesis, while also upregulating autophagy and mitophagy machinery to remove damaged mitochondria and protein aggregates. Conversely, chronic DNA damage and genotoxic stress activate PARP1, which competes with SIRT1 for the shared NAD+ pool and, when hyperactivated, depletes NAD+ and suppresses the NAD+-SIRT1-PGC-1a axis, leading to impaired mitophagy, accumulation of dysfunctional mitochondria, increased reactive oxygen species production, and loss of metabolic flexibility. This bidirectional relationship between NAD+- dependent DNA repair and metabolic homeostasis underscore the role of NAD+ in supporting genomic stability, chromatin remodeling, and epigenetic regulation, which are essential for maintaining cellular identity, and preventing age-associated transcriptional drift [9,10,13,14,16,18,19,20].
NAD+ also modulates pathways involved in stem cell exhaustion and chronic inflammation, two additional hallmarks of aging with broad implications for tissue regeneration and health span. Aged stem cells, including muscle satellite cells (MuSCs), neural stem cells, and melanocyte stem cells, exhibit NAD+ depletion, mitochondrial dysfunction, and elevated senescence markers such as CDKN1A (p21) and senescence-associated b-galactosidase, which together drive irreversible cell cycle arrest and loss of regenerative capacity. In immune cells, NAD+-modulated pathways influence the production of pro-inflammatory cytokines activation of the NLRP3 inflammasome, and the senescence-associated secretory phenotype (SASP), suggesting that NAD+ decline may contribute to “inflammaging” and the chronic low-grade inflammation that accelerates functional decline and multimorbidity. These pleiotropic and interconnected roles make NAD+ a molecular integrator through which metabolic, genotoxic, and inflammatory stressors converge to either accelerate or decelerate the aging process [9,10,13,15,16,18,21].
Preclinical Evidence for Health Span Modulation
A substantial body of preclinical research supports the concept that restoring NAD+ levels can delay age-related functional decline and extend health span in model organisms, although effects on maximal lifespan remain more variable and context-dependent. In rodent models, genetic interventions that enhance biosynthesis, such as overexpression of NAMPT or NMNAT isoforms, as well as pharmacological supplementation with NAD+ precursors including nicotinamide riboside (NR) and nicotinamide mononucleotide (NMN), have been shown to improve insulin sensitivity, enhance glucose and lipid metabolism, reduce hepatic steatosis, attenuate age-associated weight gain, and improve mitochondrial oxidative capacity and respiratory function across multiple tissues including skeletal muscle, liver, heart, and brain. Landmark studies in aged mice demonstrated that chronic NR treatment increased NAD+ levels in muscle stem cells, reduced senescence markers (γH2AX, β-galactosidase), activated the UPR^mt^ and prohibitin stress-response pathways, restored stem cell proliferative capacity and myogenic colony formation, improved muscle regeneration after injury, and extended median lifespan by approximately 5% when administered late in life. More recently, long-term NMN administration in both male and female mice has been reported to delay frailty, maintain more youthful gene expression patterns, reduce biological age as assessed by frailty indices, alter the gut microbiome (including increases in longevity-associated such as Anerotruncus colihominis) and extend median lifespan by 8.5% in female mice, although male mice showed metabolic and activity benefits without significant lifespan extension, highlighting important sex-specific differences in NAD+ metabolism and response [9,10,18,22].

Beyond effects on. Metabolic and muscle function, NAD+ restoration has demonstrated cardioprotective, neuroprotective, and regenerative benefits in preclinical models of age-related and accelerated-aging diseases. NR and NMN supplementation have been shown to delay senescence and improve function of neural stem cells and melanocyte stem cells, preserve cognitive performance in models of neurodegeneration, reduce cardiovascular dysfunction in diabetic cardiomyopathy and heart failure, and ameliorate features of accelerated aging syndromes such as muscular dystrophy (mdx mice) and progeroid models. Inhibition of the NAD+- consuming enzyme CD38, which is upregulated with age and inflammation, has similarly been shown to preserve tissue NAD+ pools, enhance mitochondrial function, and improve health span parameters, providing an alternative or complementary strategy to precursor supplementation. Importantly, while these interventions consistently extend health span, defined by improvement in physical function, metabolic health, tissue regeneration, and resistance to age-related pathology, their effects on maximal lifespan have been modest, inconsistent, or sex-specific, with some studies reporting no lifespan extension and others showing small (5-8,5%) increases in median or maximum longevity depending on dosing regimen, timing of intervention, genetic background, and sex. These findings support the concept of NAD+ as a key regulator of regenerative capacity, cellular resilience, and health span, and underscore the rationale for integrating NAD+- targeted approaches into preventive health, metabolic wellness, and regenerative medicine frameworks, while also highlighting the need for careful translation and realistic expectation setting in human applications [9,10,12,18,22,23].
NAD+ in Metabolic Health and Cardiometabolic Disease
Metabolic Regulation and Insulin Sensitivity
NAD+ is an obligate cofactor for key oxidoreductases in glycolysis, the tricarboxylic acid cycle, and the electron transport chain, making it indispensable for cellular energy production and redox homeostasis. Beyond this classical metabolic role, NAD+ functions as a regulatory signal that couple nutrient availability to transcriptional programs governing glucose and lipid metabolism, primarily via sirtuin-mediated deacetylation of metabolic transcription factors and coactivators. In particular, the NAD+- SIRT1-PGC-1a axis is a master regulator of mitochondrial biogenesis, hepatic gluconeogenesis, fatty acid oxidation, and adaptive thermogenesis, and serves as a nexus through which caloric restriction and exercise exert their beneficial metabolic effects. SIRT1 deacetylates and activates PGC-1a, which in turn drives expression of nuclear-encoded mitochondrial genes and coordinates oxidative metabolism, while mitochondrial SIRT3 deacetylates and activates enzymes involved in the TCA cycles, fatty acid oxidation, and respiratory complex assembly, thereby improving mitochondrial oxidative capacity and efficiency. Importantly, SIRT1 and SIRT3 enzymatic activity is tightly constrained by intracellular NAD+ availability, such that changes in the NAD+/ NADH ratio reflecting shifts between glycolytic and oxidative metabolism directly modulate sirtuin activity and downstream metabolic programs [24,25,26,27,28,29].

In preclinical models of obesity and metabolic syndrome, NAD+ depletion driven by reduced NAMPT expression, increased CD38 activity, and PARP hyperactivation has been shown to impair insulin sensitivity, promote hepatic steatosis, reduce skeletal muscle oxidative capacity, and accelerate adipose tissue dysfunction. Conversely, restoration of NAD+ pools via genetic overexpression of NAMPT, oral supplementation with NMN or NR, or pharmacological inhibition of CD38 has produced robust metabolic benefits including improved glucose tolerance, enhanced insulin signaling in liver and skeletal muscle, reduced hepatic triglyceride accumulation is non-alcoholic fatty liver disease (NAFLD) models, increased energy expenditure, and protection against diet-induced obesity. For example, adipocyte-specific deletion of Nampt in mice causes severe NAD+ depletion in adipose tissue, leading to multi-organ insulin resistance mediated by decreased secretion of the insulin-sensitizing adipokine adiponectin and increased circulating free fatty acids; remarkable; oral NMN supplementation fully restores adipose NAD+ levels and completely normalizes insulin resistance in these mice. Similarly, oral NMN administration to high-fat diet-fed mice improves insulin sensitivity in skeletal muscle, enhances mitochondrial oxidative metabolism, increases glucose uptake and fatty acid oxidation, and ameliorates obesity-associated metabolic dysfunction in a largely SIRT1-dependent manner. These metabolic benefits are mechanistically attributed to enhanced mitochondrial function, normalization of the NAD+/NADH redox ratio, activation of SIRT1 and SIRT3, deacetylation and activation of PGC-1a, AMPK activation, improved insulin receptor signaling, and reduced oxidative stress and inflammation. Collectively, these pathways support a strong mechanistic rationale for exploring NAD+- based interventions in insulin resistance, type 2 diabetes, NAFLD, metabolic syndrome, and obesity-related [30,31,32,33,34,35,36,37,38].
Cardiac Aging and Vascular Health
NAD+ pools decline in the aging heart and are further depleted in cardiometabolic conditions such as obesity, hypertension, and diabetes, with this decline closely associated with impaired cardiac energetic, mitochondrial dysfunction, diastolic dysfunction, increased oxidative stress, maladaptive remodeling, and heightened susceptibility to ischemic injury and heart failure. Age-related and disease-related NAD+ depletion in cardiomyocytes results from a combination of reduced biosynthetic enzyme expression (NAMPT, NRK), increased consumption by PARP1 during chronic DNA damage responses and oxidative stress, and upregulation of the NAD+ glycohydrolase CD38 in the context of inflammation and metabolic stress. This NAD+ glycohydrolase CD38 in the context of inflammation and metabolic stress. This NAD+ deficit impairs the activity of cardiac sirtuins, particularly SIRT1, SIRT3, and SIRT6 which are critical regulators of mitochondrial biogenesis, oxidative metabolism, antioxidant defenses, calcium handling, autophagy, and anti-hypertrophic signaling pathways [24,28,39,40].
NAD+- Boosting Interventions in Humans
Oral Precursors and Supplementation
Human studies investigating NAD+- boosting interventions have primarily focused on oral supplementation with NAD+ precursors, most notably nicotinamide riboside (NR), nicotinamide mononucleotide (NMN), nicotinamide (NAM), and nicotinic acid (niacin, NA), which enter distinct biosynthetic pathways and increase circulating and tissue NAD+ concentrations to varying degrees depending on dose, formulation, duration of treatment, baseline NAD+ status, and population characteristics. Systematic reviews and meta-analyses of chronic oral supplementation trials indicate that these compounds are generally well tolerated in healthy middle-aged and older adults, with the most common adverse events being mild gastrointestinal symptoms, flushing (primarily with niacin due to GPR109A receptor activation), and occasional reports of skin reactions, with NR, and NMN demonstrating more favorable safety profiles and better tolerability compared to niacin at equivalent NAD+- boosting doses. These trials consistently demonstrate biochemical efficacy, with dose-dependent increases in whole blood NAD+ levels, peripheral blood mononuclear cell (PBMC) NAD+ content, and circulating NAD+ metabolites such as nicotinamide adenine dinucleotide phosphate (NADP+), NAD+ catabolites (e.g., methyl-nicotinamide), and NAD+ pathway intermediates including nicotinic acid adenine dinucleotide (NAAD), confirming target engagement and activation of NAD+ biosynthetic pathways [41,42,43].
Despite robust evidence for biochemical efficacy and NAD+ elevation, clinical effects on robust aging endpoints and hard metabolic outcomes remain modest, heterogeneous, and largely exploratory, with most trials being small (n=10-50 participants), short in duration (2-12 weeks), and focused on surrogate biomarkers rather than disease incidence, functional decline, or mortality. Reported benefits from oral NR and NMN supplementation include improvement in some markers of cardiometabolic risk, most notably reductions in systolic and diastolic blood pressure (particularly in individuals with above-normal baseline blood pressure ³120 mmHg), decreased arterial stiffness as measured by carotid-femoral pulse wave velocity and aortic pulse wave velocity, and aortic pulse wave velocity, modest improvements in lipid profiles including increases in HDL cholesterol, reductions in inflammatory biomarkers such as C-reactive protein (CRP) and pro-inflammatory cytokines, improvements in liver function markers (alanine transaminase, ALT) , enhanced insulin sensitivity in some but not all studies, improved vascular endothelial function in hypertensive patients, increased walking distance on the 6-minutes’ walk test, enhanced muscle strength on the 30-second chair stand test, improvement in subjective fatigue, vitality, and sleep quality, and modulation of neurodegenerative biomarkers including reductions in cerebrospinal fluid (CSF) and plasma levels of amyloid-beta and tau proteins in middle-aged and older adults. Notably, a recent randomized controlled trial (RCT) in hypertensive patients (NCT04903210) demonstrated that 6 weeks of oral NMN supplementation (300 mg twice daily) significantly increased PBMC NAD+ levels, reduced systolic and diastolic blood pressure by approximately 5-7 mmHg, improved endothelial-dependent vasodilation (acetylcholine-induced vasorelaxation, reduced arterial stiffness and lowered plasma inflammatory markers, providing some of the strongest human evidence to date for clinically meaningful cardiovascular benefits [42,43,44,45,46,47].
However, many of these findings remain preliminary, and some trials report neutral or negative results, particularly in healthy individuals with normal baseline cardiometabolic parameters, underscoring the importance of patient selection, baseline NAD+ status, and phenotypic heterogeneity in determining response to NAD+ supplementation. Furthermore, while short-term trials (< 12 weeks) show favorable safety profiles, large-scale, long-term randomized trials are urgently needed to determine whether chronic NAD+ augmentation meaningfully alters disease incidence, functional decline, disability, or all-cause mortality in aging populations, to identify rare adverse events that may emerge only with prolonged use or in high-risk subgroups (e.g., individuals with cancer, autoimmune disease, or mitochondrial (disorders) and to establish optimal dosing regimens, timing of intervention (midlife vs. late life), and combination strategies with other geroscience interventions. Importantly, head-to-head comparisons of different NAD+ precursors, tissue-specific NAD+ profiling, and isotope-tracing studies to quantify NAD+ flux dynamics in humans are lacking, limiting conclusions about which precursors are most effective for specific tissues, disease contexts, or therapeutic goals [41,42,43,44,46].
Parenteral, Topical and Regenerative Applications
Beyond oral supplementation, NAD+ and its precursors are increasingly being explored in intravenous (IV) infusions, intramuscular (IM) injections, subcutaneous formulations, topical products, and peri-procedural protocols for regenerative medicine, aesthetic medicine, surgical recovery, and wellness applications, although the clinical evidence base for these modalities remains substantially more limited than for oral supplementation. Proponents of IV NAD+ therapy argue that parenteral administration bypasses first-pass hepatic metabolism and delivers higher, more sustained concentrations of NAD+ directly to the systemic circulation and tissues, resulting in more rapid and pronounced effects on cellular energy production, mitochondrial function, cognitive clarity, physical recovery, and subjective well-being compared to oral routes. Proposed benefits of IV NAD+ infusions include enhanced cellular energy and ATP production, improved mental clarity focus, and emotional stability, reduced fatigue and burnout in high-stress or high-performing individuals, accelerated recovery after aesthetic and surgical procedures (e.g., microneedling, laser resurfacing, radiofrequency treatments, facelifts, liposuction), support for collagen production and skin firmness, enhanced skin radiance and hydration, reduced oxidative stress and inflammation contributing to skin aging, and promotion of a balanced, revitalized appearance with smoother texture [4].
In regenerative and aesthetic contexts, NAD+ is also being administered as a concentrated topical application post-operatively to improve wound healing and as an injectable in combination with microneedling, micro-coring (e,g,m Ellacor), platelet-rich plasma (PRP), exosome therapy, and other regenerative modalities, with the rationale that optimizing NAD+ status enhances cellular resilience, mitochondrial biogenesis, and tissue repair capacity, thereby supporting faster healing, reducing inflammation, and optimizing long-term aesthetic outcomes in mature or photoaged skin where mitochondrial function and cellular turnover are often compromised. Pre-treatment NAD+ infusions are hypothesized to prepare the skin and tissues for invasive or energy-based interventions by increasing cellular energy reserves, antioxidant defenses, and stress resistance, while post-treatment use is proposed to support faster recovery, reduce downtime, minimize inflammation, and enhance collages synthesis and remodeling [4].
However, rigorous, controlled clinical data supporting these applications remain limited, and many claims derive from preclinical work, mechanistic extrapolations, uncontrolled observational reports, or testimonial evidence rather than from randomized, placebo-controlled trials with objective endpoints. IV NAD+ therapy requires more time (infusions often take 2-4 hours) and clinical oversight compared to oral or IM routes, and potential risks including infusion reactions, electrolyte imbalances, and interactions with other medications have not been systematically evaluated in large cohorts or long-term follow-up studies. Moreover, NAD+ itself is a large, charged molecule with limited membrane permeability, raising questions about whether direct IV or topical NAD+ administration is more effective than precursor-based strategies that rely on intracellular conversion to NAD+ via endogenous biosynthetic pathways, and whether the acute, transient elevation of circulating NAD+ achieved by IV infusion translates into sustained tissue NAD+ accumulation and functional benefits. Translating these concepts into standard practice will require mechanistic human studies employing tissue biopsies, advanced imaging, and multi-omics profiling to determine whether and how parenteral NAD+ modulates tissue-level NAD+ pools, mitochondrial function, gene expression, and repair processes, as well as randomized controlled trials that integrate NAD+, as well as randomized controlled trials that integrate NAD+ modulation into broader peri-procedural protocols and lifestyle interventions (e.g., exercise, sleep optimization, nutritional support) to determine additive or synergistic effects on surgical outcomes, recovery time, complication rates, and patient-reported quality of life. Until such data are available, parenteral and topical NAD+ applications should be considered investigational and used with informed consent, transparent communication of uncertainties, and close monitoring for safety and efficacy signals [4,42].
Safety, Limitations and Controversies
Short-to medium-term human trials of oral NR and NMN (typically 2-12 months, doses ~250-2,000 mg/day) have generally demonstrated a favorable safety profile, with mild gastrointestinal symptoms (nausea, diarrhea, abdominal discomfort), headache, flushing and pruritus among the most frequently reported adverse events and with low discontinuation rates. Laboratory parameters including liver enzymes, renal function, hematologic indices, and glycemic markers have remained stable in most studies, and serious adverse events have been rare and not clearly attributable to NAD+ precursors, leading current reviews to classify NR and NMN as well tolerated in healthy and cardiometabolic-risk populations over the short term, nonetheless, concerns persist about potential long-term risks, particularly in individuals at risk for or living with cancer, because NAD+ supports and repair, mitochondrial function, and anabolic metabolism processes that can be co-opted by malignant cells, raising the theoretical possibility that chronic NAD+ elevation could promote tumor initiation, progression, or resistance to therapy in certain contexts. In addition, supra-physiologic NAD+ levels or sustained activation of NAD+- consuming enzymes (e.g., PARPs, sirtuins, cd38) could, in principle, perturb cellular redox balance, alter innate and adaptive immune responses, and interfere with circadian and metabolic rhythms in ways that have not yet been fully characterized in humans, underscoring the need for robust dose-finding, pharmacokinetic-pharmacodynamic modeling, and long-duration safety studies that include high-risk subgroups. Until such data are available, NAD+- boosting interventions are best regarded as experimental adjuncts within preventive and aging-wellness programs rather than established anti-aging therapies, and should be used with careful patient selection, documentation of baseline risk, and transparent discussion of uncertainties [2,10,41,42,43,44,48].
Despite compelling mechanistic and preclinical data, including consistent improvements in metabolic health, cardiovascular and neurodegenerative phenotypes, and multiple dimensions of health span in rodent models, definitive evidence that NAD+- boosting strategies extend human health span or lifespan is currently lacking. Even in animal studies, benefits on health span do not uniformly translate into increased maximal lifespan, with several NMN and NR interventions producing modest or sex-specific gains in median lifespan (on the order of 5-8.5%) or no significant lifespan extension despite clear functional improvements, highlighting context dependence and the limits of extrapolating from model organisms to humans. Interindividual heterogeneity in baseline NAD+ status, genetic variants affecting NAD+ biosynthetic and consuming enzymes, microbiome composition (which influences precursor metabolism), dietary patterns, physical activity, medication use, and comorbidities likely modulates both the magnitude and direction of response to NAD+- boosting therapies, supporting a shift toward personalized, biomarker-guided approaches rather than uniform supplementation strategies. At the same time, there is a growing disconnect between aggressive commercial claims particularly in wellness, biohacking, and aesthetic markets and the current strength of clinical evidence which consist largely of small, short-term trials focused on surrogate markers; this gap risks eroding trust and may lead to misuse or overuse if expectations are not aligned with available data. Moving NAD+- targeted interventions from hype to evidence-based practice will require standardized and validated biomarkers of NAD+ status and flux (e.g., blood and tissue NAD+ metabolomics), harmonized clinical endpoints that capture both health span and disease-specific outcomes, and the integration of multi-omics, digital phenotyping, and AI-driven longitudinal monitoring to identify responders, quantify benefit-risk profiles, and optimize combination regimens with lifestyle and other geroscience interventions [2,4,9,10,13,15,18,42,43,48].
Future Directions for Metabolic Aging Wellness
Future application of NAD⁺-targeted strategies in metabolic and aging wellness is expected to focus on individuals with demonstrable NAD⁺ depletion, clustered cardiometabolic risk, or distinct aging phenotypes such as frailty, sarcopenia, or early vascular aging. This will likely involve combining practical NAD⁺ metrics with metabolic, inflammatory, and epigenetic biomarkers and increasingly, wearable-derived digital phenotypes to refine risk stratification and monitor responses over time. Rational combination regimens that pair NAD⁺ restoration with lifestyle interventions, caloric restriction mimetics, senotherapeutics, or other geroscience-guided therapies are being explored to achieve synergistic health span gains rather than relying on NAD⁺ alone. Embedding these measurements and interventions into AI-enabled, longitudinal health platforms may enable adaptive, data-driven personalization of metabolic and aging wellness programs as clinical evidence evolves [9,13,19].
Conclusion
NAD+ has emerged as a central integrator of metabolic regulation and aging biology, connecting energy metabolism, DNA repair, cellular stress responses, and inflammatory signaling across multiple tissues. Age-related declines in NAD+ availability appear to contribute to cardiometabolic disease, neurodegeneration, and other chronic conditions, while preclinical studies consistently demonstrate that restoring NAD+ levels can improve health span-related phenotypes, including metabolic flexibility, cardiovascular function, and tissue regenerative capacity.
Human trials of NAD+- boosting interventions confirm biochemical efficacy, robust increases in circulating and cellular NAD+ pools and generally favorable short-term safety, yet provide only preliminary and heterogeneous evidence for clinically meaningful benefits on aging and metabolic outcomes. this evidentiary gap underscores the need for larger, longer-duration, and better- phenotype studies that incorporate standardized NAD⁺ biomarkers, hard clinical endpoints, and stratification by baseline risk and molecular phenotype.
Within metabolic and aging wellness programs, NAD⁺ is therefore best viewed as a promising mechanistic target and potential adjunct to foundational lifestyle and risk-factor interventions rather than a stand-alone anti-aging therapy. Responsible clinical translation will require rigorous biomarker integration, realistic expectation-setting, transparent communication of uncertainties, and ongoing clinical research to define which individuals benefit most, at what doses, and in which combinatorial regimens NAD⁺-targeted strategies can safely and meaningfully contribute to health span extension.
Reference
- Zhang W, Ren H, Chen W, Hu B, Feng C, Li P, et al. Nicotinamide phosphoribosyltransferase in NAD+ metabolism: physiological and pathophysiological implications. Cell Death Discovery [Internet]. 2025 Aug 8 [cited 2025 Dec 7];11(1):371–1. Available from: https://www.nature.com/articles/s41420-025-02672-w
- Xie N, Zhang L, Gao W, Huang C, Huber PE, Zhou X, et al. NAD+ metabolism: pathophysiologic mechanisms and therapeutic potential. Signal Transduction and Targeted Therapy [Internet]. 2020 Oct 7;5(227). Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7539288/
- Su M, Qiu F, Li Y, Che T, Li N, Zhang S. Mechanisms of the NAD+ salvage pathway in enhancing skeletal muscle function. Frontiers in Cell and Developmental Biology [Internet]. 2024 Sep 20;12. Available from: https://pdfs.semanticscholar.org/1eaf/17f8c875b030dd8361af378858fd25426884.pdf
- Conlon N, Ford D. A systems-approach to NAD+ restoration. Biochemical Pharmacology. 2022 Apr;198:114946.
- Yoshino J, Baur JA, Imai S. NAD+ Intermediates: The Biology and Therapeutic Potential of NMN and NR. Cell Metabolism [Internet]. 2018 Mar;27(3):513–28. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5842119/
- Yaku K, Palikhe S, Iqbal T, Hayat F, Watanabe Y, Fujisaka S, et al. Nicotinamide riboside and nicotinamide mononucleotide facilitate NAD synthesis via enterohepatic circulation. Science Advances. 2025 Mar 21;11(12).
- Mascio J. Overview of NAD+ and Its Role in the BodyScientists found that there was a fundamental molecule called nico… [Internet]. Goldman Laboratories. 2025 [cited 2026 Jan 15]. Available from: https://goldmanlaboratories.com/blogs/blog/nad-pathways
- CreativeWork [Internet]. Wikipathways.org. WikiPathways; 2026 [cited 2026 Jan 15]. Available from: https://pfocr.wikipathways.org/figures/PMC5967641__nihms954031f1.html
- Imai S, Guarente L. NAD+ and sirtuins in aging and disease. Trends in Cell Biology. 2014 Aug;24(8):464–71.
- Covarrubias AJ, Perrone R, Grozio A, Verdin E. NAD+ metabolism and its roles in cellular processes during ageing. Nature Reviews Molecular Cell Biology. 2020 Dec 22;22(2):119–41.
- Nicotinamide Adenine Dinucleotide [Internet]. Bpsbioscience.com. 2015. Available from: https://bpsbioscience.com/nad-plus-nicotinamide-adenine-dinucleotide
- Zeidler JD, Hogan KA, Agorrody G, Peclat TR, Kashyap S, Kanamori KS, et al. The CD38 glycohydrolase and the NAD sink: implications for pathological conditions. AJP Cell Physiology. 2022 Feb 9;322(3):C521–45.
- Sanada F, Hayashi S, Morishita R. Targeting the hallmarks of aging: mechanisms and therapeutic opportunities. Frontiers in Cardiovascular Medicine [Internet]. 2025 Jul 1;12. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC12259695/
- Lee IH, Cao L, Mostoslavsky R, Lombard DB, Liu J, Bruns NE, et al. A role for the NAD-dependent deacetylase Sirt1 in the regulation of autophagy. Proceedings of the National Academy of Sciences of the United States of America [Internet]. 2008 Mar 4;105(9):3374–9. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2265142/
- Delrue C, Reinhart Speeckaert, Speeckaert MM. Rewinding the Clock: Emerging Pharmacological Strategies for Human Anti-Aging Therapy. International Journal of Molecular Sciences [Internet]. 2025 Sep 25 [cited 2026 Jan 15];26(19):9372–2. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC12524491/
- Ur Rahman S, Qadeer A, Wu Z. Role and Potential Mechanisms of Nicotinamide Mononucleotide in Aging. Aging and Disease [Internet]. 2023 Jul 27; Available from: https://pubmed.ncbi.nlm.nih.gov/37548938/
- Hipkiss AR. Mitochondrial dysfunction, proteotoxicity, and aging: causes or effects, and the possible impact of NAD+-controlled protein glycation. Advances in clinical chemistry [Internet]. 2010;50:123–50. Available from: https://pubmed.ncbi.nlm.nih.gov/20521444/
- Zhang H, Ryu D, Wu Y, Gariani K, Wang X, Luan P, et al. NAD+ repletion improves mitochondrial and stem cell function and enhances life span in mice. Science [Internet]. 2016 Apr 28;352(6292):1436–43. Available from: https://science.sciencemag.org/content/352/6292/1436.long
- Delrue C, Reinhart Speeckaert, Speeckaert MM. Rewinding the Clock: Emerging Pharmacological Strategies for Human Anti-Aging Therapy. International Journal of Molecular Sciences [Internet]. 2025 Sep 25;26(19):9372–2. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC12524491/
- Wan W, Hua F, Fang P, Li C, Deng F, Chen S, et al. Regulation of Mitophagy by Sirtuin Family Proteins: A Vital Role in Aging and Age-Related Diseases. Frontiers in Aging Neuroscience. 2022 May 9;14.
- Lagunas-Rangel FA. Sirtuins in mitophagy: key gatekeepers of mitochondrial quality. Molecular and Cellular Biochemistry [Internet]. 2025 Jul 24 [cited 2026 Jan 15];480(12):5877–96. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC12594717/
- Do NADBoosters Work? What the Research Says About NR and NMN for Aging, Cognition & Muscle | Healthspan [Internet]. Healthspan. Healthspan website; 2025 [cited 2026 Jan 15]. Available from: https://www.gethealthspan.com/research/article/nad-boosters
- Conlon NJ. The Role of NAD+ in Regenerative Medicine. Plastic & Reconstructive Surgery. 2021 Sep 28;150:41S48S.
- Jahan F, Bagchi RA. Enhancing NAD+ Metabolome in Cardiovascular Diseases: Promises and Considerations. Frontiers in Cardiovascular Medicine. 2021 Aug 27;8.
- Welcome To Zscaler Directory Authentication [Internet]. Nih.gov. 2026 [cited 2026 Jan 15]. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC8123861/
- Cantó C, Auwerx J. PGC-1α, SIRT1 and AMPK, an energy sensing network that controls energy expenditure. Current Opinion in Lipidology [Internet]. 2009 Apr;20(2):98–105. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3627054/
- Yamaguchi S, Yoshino J. Adipose tissue NADbiology in obesity and insulin resistance: From mechanism to therapy. BioEssays [Internet]. 2017 Mar 15;39(5):1600227. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5469033/
- Kane AE, Sinclair DA. Sirtuins and NAD+in the Development and Treatment of Metabolic and Cardiovascular Diseases. Circulation Research. 2018 Sep 14;123(7):868–85.
- Cantó C, Auwerx J. Targeting Sirtuin 1 to Improve Metabolism: All You Need Is NAD+? Desvergne B, editor. Pharmacological Reviews. 2011 Nov 21;64(1):166–87.
- Yamaguchi S, Yoshino J. Adipose tissue NADbiology in obesity and insulin resistance: From mechanism to therapy. BioEssays [Internet]. 2017 Mar 15;39(5):1600227. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5469033/
- Welcome To Zscaler Directory Authentication [Internet]. Nih.gov. 2026. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC8123861/
- Kane AE, Sinclair DA. Sirtuins and NAD+in the Development and Treatment of Metabolic and Cardiovascular Diseases. Circulation Research. 2018 Sep 14;123(7):868–85.
- Cantó C, Auwerx J. Targeting Sirtuin 1 to Improve Metabolism: All You Need Is NAD+? Desvergne B, editor. Pharmacological Reviews. 2011 Nov 21;64(1):166–87.
- Lu X, Yang R, Chen Y, Chen D. NAD metabolic therapy in metabolic dysfunction-associated steatotic liver disease: Possible roles of gut microbiota. iScience. 2024 Feb 9;27(3):109174–4.
- Majeed Y, Halabi NM, Engelke R, Hina Sarwath, Al-Noubi MN, Choi S, et al. NAD+ boosting by oral nicotinamide mononucleotide administration regulates key metabolic and immune pathways through SIRT1 dependent and independent mechanisms to mitigate diet-induced obesity and dyslipidemia in mice. 2025 Nov 10 [cited 2026 Jan 15]; Available from: https://elifesciences.org/reviewed-preprints/109214v1
- Yoshino M, Yoshino J, Kayser BD, Patti G, Franczyk MP, Mills KF, et al. Nicotinamide mononucleotide increases muscle insulin sensitivity in prediabetic women. Science [Internet]. 2021 Apr 22;372(6547). Available from: https://science.sciencemag.org/content/early/2021/04/21/science.abe9985
- Dong M, Wang S, Pei Z. Mechanism of CD38 via NADin the Development of Non-alcoholic Fatty Liver Disease. International Journal of Medical Sciences. 2023;20(2):262–6.
- Zhang W, Ren H, Chen W, Hu B, Feng C, Li P, et al. Nicotinamide phosphoribosyltransferase in NAD+ metabolism: physiological and pathophysiological implications. Cell Death Discovery [Internet]. 2025 Aug 8 [cited 2025 Dec 7];11(1):371–1. Available from: https://www.nature.com/articles/s41420-025-02672-w
- Abdellatif M, Sedej S, Kroemer G. NAD Metabolism in Cardiac Health, Aging, and Disease. Circulation. 2021 Nov 30;144(22):1795–817.
- Yuan Y, Liang B, Liu XL, Liu WJ, Huang BH, Yang SB, et al. Targeting NAD+: is it a common strategy to delay heart aging? Cell Death Discovery [Internet]. 2022 Apr 26 [cited 2026 Jan 15];8(1). Available from: https://www.nature.com/articles/s41420-022-01031-3
- Fabiana G, Glaucia Maria Pastore. NAD+ Precursors Nicotinamide Mononucleotide (NMN) and Nicotinamide Riboside (NR): Potential Dietary Contribution to Health. Current Nutrition Reports. 2023 Jun 5;
- Khatri S, Abdellatif M, Sedej S. Clinical evidence for the use of NAD+ precursors to slow aging. 2025 Jan 1; Available from: https://www.researchgate.net/publication/397665886_Clinical_evidence_for_the_use_of_NAD_precursors_to_slow_aging
- Freeberg KA, Udovich CC, Martens CR, Seals DR, Craighead DH. Dietary Supplementation With NAD+-Boosting Compounds in Humans: Current Knowledge and Future Directions. The Journals of Gerontology Series A, Biological Sciences and Medical Sciences [Internet]. 2023 Dec 1;78(12):2435–48. Available from: https://pubmed.ncbi.nlm.nih.gov/37068054/
- Martens CR, Denman BA, Mazzo MR, Armstrong ML, Reisdorph N, McQueen MB, et al. Chronic nicotinamide riboside supplementation is well-tolerated and elevates NAD+ in healthy middle-aged and older adults. Nature Communications. 2018 Mar 29;9(1).
- Yu B, Jing X, Jia L, Wang M, Liu L, Ping S, et al. The versatile multi-functional substance NMN: its unique characteristics, metabolic properties, pharmacodynamic effects, clinical trials, and diverse applications. Frontiers in Pharmacology. 2024 Oct 1;15.
- Qiu Y, Xu S, Chen X, Wu X, Zhou Z, Zhang J, et al. NAD+ exhaustion by CD38 upregulation contributes to blood pressure elevation and vascular damage in hypertension. Signal Transduction and Targeted Therapy. 2023 Sep 18;8(1).
- Lei L, Zhang X, Lin J, Liang Q, Sohouli MH, Izze da Silva Magalhães E, et al. Effects of NAD+ precursors on blood pressure, C-reactive protein concentration and carotid intima-media thickness: A meta-analysis of randomized controlled trials. European journal of clinical investigation [Internet]. 2023 Dec;53(12):e14078. Available from: https://pubmed.ncbi.nlm.nih.gov/37593976/
- Iqbal T, Nakagawa T. The therapeutic perspective of NAD+ precursors in age-related diseases. Biochemical and Biophysical Research Communications. 2024 Apr 1;702:149590–0.